Hydrate K VH values (defined as a component’s mole fraction divided by that in the hydrate) for each gas component are used to determine a hydrate dewpoint for a gas of constant composition.
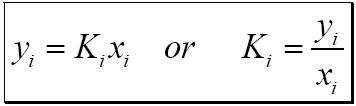
At temperatures below 277 K, pressures below the 1.2 MPa average are required. Of the 20 gases, the lowest formation pressure was 0.67 MPa for a gas with 7.0 mol% C 3H 8, while the highest value was 2.00 MPa for a gas with 1.8 mol% C 3H 8. Hydrate formation data at 277 K were averaged for 20 natural gases, and the average formation pressure was 1.2 MPa.
#Depriester chart propane free
For example, at a typical seafloor temperature of 277 K, hydrates will form in a natural gas system if free water is available and the pressure is greater than 1.2 MPa.


A clear, prescriptive method for constructing the hydrate flash program has recently been published. The basis for both program types is a hydrate equation of state (EOS). State-of-the-art programs are transitioning to the flash/Gibbs free-energy type. Of these two program types, the flash/Gibbs type is gaining pre-eminence because its predictions are available in the phase diagram interior (where many systems operate), whereas the incipient type provides the pressure/temperature (P/T) points of hydrate initiation.

